The battle between plaque and tartar represents one of the most fundamental challenges in oral health care. While many dental professionals understand these as distinct entities, the complex process of biofilm formation and mineralisation reveals why conventional mechanical cleaning often falls short. Understanding how microscopic bacterial communities transform from soft, removable plaque into hardened tartar deposits provides crucial insights for developing more effective treatment protocols and patient education strategies.

This progression from initial bacterial adhesion to established periodontal disease follows predictable pathways, yet individual variations in biofilm development create unique challenges for each patient. Modern research reveals that even the most thorough brushing removes only 65% of plaque, leaving substantial bacterial populations to mature and mineralise. The implications extend far beyond oral health, with emerging evidence linking persistent biofilms to cardiovascular disease, diabetes, and neurological conditions.
What is dental plaque and how does biofilm formation begin?
Dental plaque represents a complex microbial ecosystem that begins forming within minutes of thorough cleaning. This sticky biofilm consists of over 700 different bacterial species embedded in a protective matrix of proteins, polysaccharides, and extracellular DNA. The formation process starts when salivary proteins create a conditioning film on clean tooth surfaces, providing attachment sites for primary colonising bacteria.
The initial colonisers include Streptococcus sanguis, Streptococcus mitis, and Actinomyces species, which possess specific adhesins that allow them to bind to tooth enamel and exposed root surfaces. Within 2–4 hours, these pioneer bacteria begin multiplying and secreting extracellular polymeric substances that form the foundation of the biofilm matrix. This rapid establishment explains why teeth can feel rough or fuzzy just hours after professional cleaning.
Secondary colonisers then attach to the initial bacterial layer through co-aggregation mechanisms. These include more pathogenic species like Fusobacterium nucleatum, which acts as a bridge organism, facilitating attachment of late colonisers such as Porphyromonas gingivalis and Tannerella forsythia. The biofilm reaches maturity within 7–14 days, creating a complex three-dimensional structure with water channels and distinct microenvironments.
How plaque transforms into tartar through mineralisation
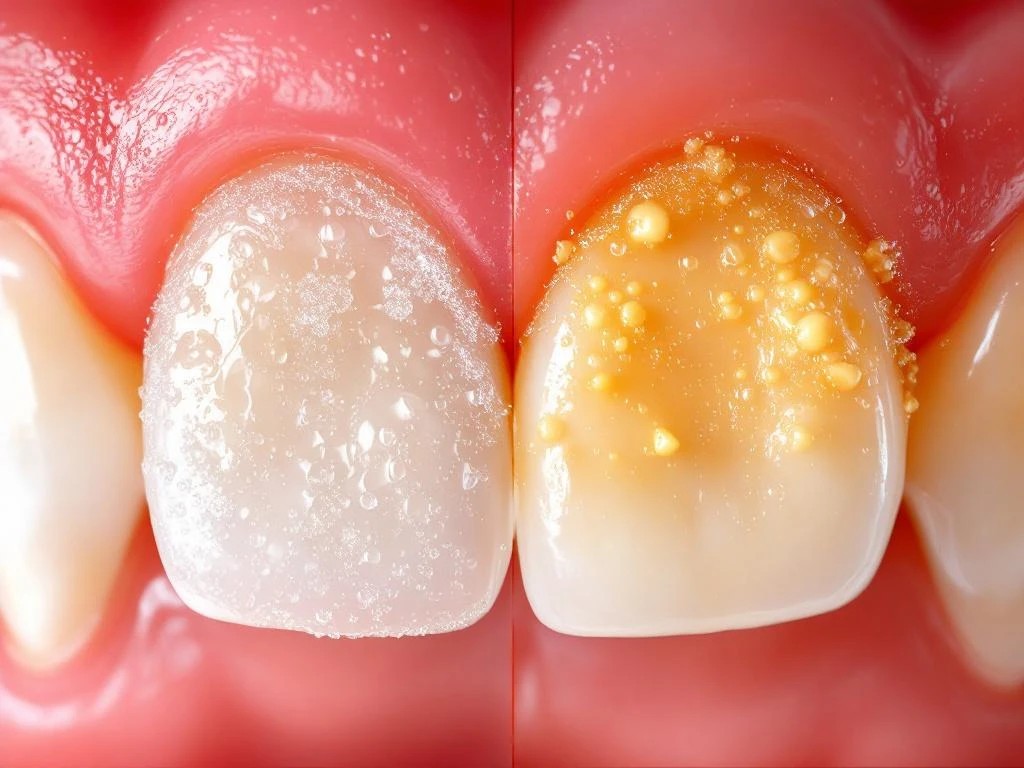
The transformation of soft plaque into hardened tartar occurs through a mineralisation process driven by salivary and gingival crevicular fluid components. Calcium and phosphate ions from saliva precipitate within the biofilm matrix, forming hydroxyapatite and other calcium phosphate crystals. This process typically begins within 2–14 days of plaque formation, though individual variations in saliva composition significantly affect the timeline.
Supragingival tartar formation occurs above the gum line, primarily influenced by saliva mineral content and pH levels. Areas near salivary gland openings, particularly the lingual surfaces of lower incisors and buccal surfaces of upper molars, show accelerated tartar formation. The mineralisation process creates a rough, porous surface that provides ideal retention sites for additional bacterial accumulation.
Subgingival tartar develops below the gum line in periodontal pockets, where gingival crevicular fluid provides the mineral source. This type appears darker due to incorporation of blood products and tends to be more tenacious than supragingival deposits. Once formed, tartar cannot be removed through mechanical brushing or flossing and requires professional scaling with ultrasonic or hand instruments. The rough surface texture of tartar facilitates rapid plaque re-accumulation, creating a cycle of bacterial retention and further mineralisation.
Why biofilm protects harmful bacteria from conventional cleaning
The biofilm matrix creates a sophisticated protective barrier that shields pathogenic bacteria from mechanical disruption and antimicrobial agents. Extracellular polymeric substances form up to 85% of the biofilm structure, creating a gel-like environment that restricts penetration of antibiotics, antiseptics, and immune system components. This matrix consists of polysaccharides, proteins, lipids, and extracellular DNA that bind together to form a cohesive protective layer.
Within mature biofilms, bacteria exhibit dramatically increased resistance to antimicrobial agents compared to their planktonic counterparts. This resistance stems from multiple mechanisms, including reduced metabolic activity in deeper biofilm layers, enzymatic degradation of antimicrobials, and altered gene expression patterns. The biofilm architecture creates oxygen and nutrient gradients that allow anaerobic pathogens to thrive in protected microenvironments.
Bacterial communication through quorum sensing enables coordinated responses to environmental threats. When biofilms detect mechanical or chemical challenges, they can rapidly alter their structure, increase matrix production, or release planktonic cells to establish new colonies elsewhere. This sophisticated defence system explains why conventional brushing and antimicrobial mouth rinses often fail to eliminate established plaque biofilms, particularly in areas with limited access such as interproximal spaces and subgingival regions.
Environmental factors that accelerate biofilm development
Saliva composition plays a crucial role in biofilm formation rates, with variations in mineral content, pH, and protein concentrations significantly affecting plaque development. Individuals with higher salivary calcium and phosphate levels experience accelerated tartar formation, while those with reduced saliva flow (xerostomia) face increased biofilm accumulation due to decreased natural cleansing mechanisms. Saliva pH below 5.5 creates acidic conditions that promote cariogenic bacterial growth and enamel demineralisation.
Dietary factors substantially influence biofilm composition and growth patterns. Frequent consumption of fermentable carbohydrates provides substrate for acid-producing bacteria, creating localised pH drops that favour pathogenic species. Sticky foods and sugary beverages create prolonged bacterial feeding opportunities, while acidic drinks can soften enamel and create retention sites for initial bacterial adhesion.
Systemic health conditions significantly impact biofilm development rates. Diabetes mellitus alters immune function and creates elevated glucose levels in gingival crevicular fluid, promoting bacterial growth. Hormonal changes during pregnancy, menstruation, and menopause affect gingival tissue response and can accelerate biofilm-induced inflammation. Medications that cause dry mouth, including antihistamines, antidepressants, and blood pressure medications, reduce saliva’s protective effects and allow enhanced bacterial colonisation.
From biofilm to periodontal disease progression
Persistent biofilms trigger inflammatory cascades that progress from reversible gingivitis to destructive periodontitis through well-documented pathways. Initial gingival inflammation occurs when bacterial toxins and metabolic products penetrate gingival tissues, activating innate immune responses. Lipopolysaccharides from gram-negative bacteria stimulate inflammatory mediator release, including interleukin-1β, tumour necrosis factor-α, and prostaglandin E2.
The progression from gingivitis to periodontitis involves complex host–microbe interactions in which the inflammatory response intended to combat bacterial invasion ultimately causes tissue destruction. Matrix metalloproteinases activated during chronic inflammation degrade collagen fibres supporting gingival attachment, while osteoclast activation leads to alveolar bone resorption. This creates deeper periodontal pockets that provide protected environments for anaerobic pathogenic bacteria.
Systemic health implications of chronic periodontal inflammation extend far beyond oral tissues. Bacteraemia from daily activities like tooth brushing allows oral pathogens to enter systemic circulation, potentially contributing to cardiovascular disease, diabetes complications, and adverse pregnancy outcomes. Research indicates that untreated gingivitis doubles stroke risk in individuals under 50, while periodontal pathogens have been identified in atherosclerotic plaques and brain tissue, suggesting direct pathogenic roles in systemic disease development.
Modern approaches to biofilm management and prevention
Contemporary biofilm management strategies combine mechanical disruption with targeted antimicrobial approaches that address the limitations of conventional oral hygiene methods. Professional scaling and root planing remain the gold standard for removing mineralised deposits, while adjunctive chemical agents such as chlorhexidine provide temporary bacterial reduction. However, broad-spectrum antiseptics can disrupt beneficial oral microflora, potentially leading to microbial imbalances.
Emerging light-assisted antibacterial technologies represent a significant advancement in targeted biofilm management. Photodynamic therapy utilises light-activated compounds that selectively bind to bacterial biofilms, creating localised antimicrobial effects without disrupting healthy tissue or beneficial bacteria. These approaches can eliminate 99.998% of harmful plaque bacteria while supporting healthy oral bacterial diversity, addressing the limitations of conventional mechanical and chemical methods.
The Lumoral system combines dual-light technology with photosensitising agents to target biofilm bacteria with microscopic precision. This approach uses 405 nm blue light for direct bacterial inactivation and 810 nm near-infrared light for tissue support, providing comprehensive biofilm management that complements traditional home hygiene routines. Such targeted approaches offer particular benefits for patients with compromised manual dexterity, high tartar formation rates, or areas that are difficult to clean mechanically.
Integration of these advanced technologies with conventional oral care creates comprehensive prevention strategies that address biofilm formation at multiple stages. Regular professional maintenance, optimised home hygiene techniques, and targeted antimicrobial interventions work synergistically to maintain healthy oral ecosystems while preventing the progression from plaque to tartar and subsequent periodontal disease. For more information about science-backed oral hygiene innovations, dental professionals can explore evidence-based approaches that enhance patient outcomes through improved biofilm management protocols.